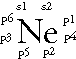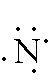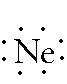Examples: Lithium: 1s2 2s1 -- 2 is the outer energy level. Since it is 2s1, there is only 1 electron in the outer energy level. Thus the Lewis Dot Diagram should look like:
Beryllium: 1s2 2s2 -- 2 is the outer energy level. Since it is 2s2, there are 2 electrons in the outer energy level. Remember, both of these electrons in the outer energy level are in the s-orbital. Thus they should be written together and the Lewis Dot Diagram should look like:
Boron: 1s2 2s2 2p1 -- 2 is the outer energy level. Since it is 2s2 and 2p1, there are 3 electrons in the outer energy level. Remember, both of the electrons in the s-orbital have to be written together. The 1 p-orbital electron can go in any empty space. The Lewis Dot Diagram should look like:
Carbon: 1s2 2s2 2p2 -- 2 is the outer energy level. Since it is 2s2 and 2p2, there are 4 electrons in the outer energy level. Remember, both of the electrons in the s-orbital have to be written together. The 2 p-orbital electrons will each fill an empty space before they pair up. The Lewis Dot Diagram should look like:
Nitrogen: 1s2 2s2 2p3 -- -- 2 is the outer energy level. Since it is 2s2 and 2p3, there are 5 electrons in the outer energy level. Remember, both of the electrons in the s-orbital have to be written together. The 3 p-orbital electrons will each fill an empty space before they pair up. The Lewis Dot Diagram should look like:
Oxygen: 1s2 2s2 2p4 -- 2 is the outer energy level. Since it is 2s2 and 2p4, there are 6 electrons in the outer energy level. Remember, both of the electrons in the s-orbital have to be written together. The 4 p-orbital electrons will each fill an empty space before they pair up. Since there are only 3 empty spaces but 4 p electrons, two of the electrons must double up. The Lewis Dot Diagram should look like:
Fluorine: 1s2 2s2 2p5 -- 2 is the outer energy level. Since it is 2s2 and 2p5, there are 7 electrons in the outer energy level. Remember, both of the electrons in the s-orbital have to be written together. The 5 p-orbital electrons will each fill an empty space before they pair up. Since there are only 3 empty spaces but 5 p electrons, four of the electrons must double up. The Lewis Dot Diagram should look like:
Neon: 1s2 2s2 2p6 -- 2 is the outer energy level. Since it is 2s2 and 2p6, there are 8 electrons in the outer energy level. Remember, both of the electrons in the s-orbital have to be written together. The 6 p-orbital electrons will each fill an empty space before they pair up. Since there are only 3 empty spaces but 6 p electrons, all six of the electrons must double up. The Lewis Dot Diagram should look like:
In
summary, elements in:
|